The Defense Advanced Research Projects Agency (DARPA) has awarded a cooperative agreement to Massachusetts General Hospital for research and development supporting the Safe Genes program.
The Safe Genes program aims to deliver novel biological capabilities to facilitate the safe and expedient pursuit of advanced genome editing applications, while also providing the tools and methodologies to mitigate the risk of unintentional consequences or intentional misuse of these technologies.
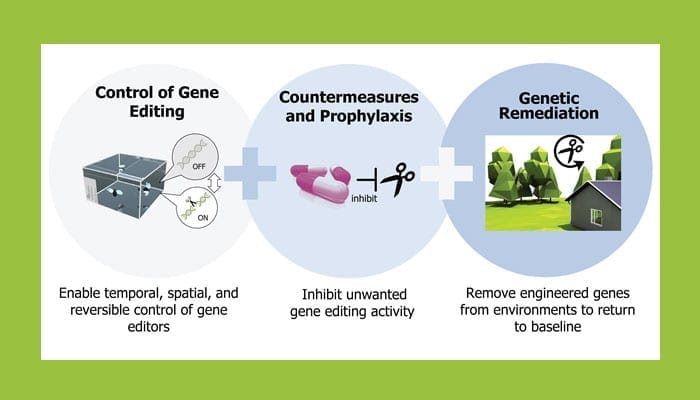
There are three primary technical focus areas within the Safe Genes program to help bridge the gap between the current technological landscape and the future transformative applications of genome editing tools and engineered organisms. First, researchers will design and develop the genetic circuitry and genome editing machinery for robust, spatial, temporal, and reversible control of genome editing activity in living systems.
The second technical focus area will develop small molecules and/or molecular strategies to provide prophylactic and treatment solutions that prevent or limit genome editing activity and protect the genome integrity of organisms and populations.
Finally, researchers will develop “genetic remediation” strategies that eliminate unwanted engineered genes from a broad range of complex population and environmental contexts to restore systems to functional and genetic baseline states. Overall the program aims to create a layered, modular, and adaptable solution set to access translational applications of genome editing technologies while protecting against intentional or accidental misuse of these tools.
The contract has a ceiling value of $11,000,660. The scope of Massachusetts General Hospital’s work under the Safe Gene program is unspecified.
Work will be performed in Boston, Massachusetts (51 percent); London, United Kingdom (24 percent); Raleigh, North Carolina (8 percent); and Perugia, Italy (17 percent), with an expected completion date of April 2021. Fiscal 2016 funds in the amount of $987,271 are being obligated at time of award.
This was a competitive acquisition and 17 offers were received. The Defense Advanced Research Projects Agency, Arlington, Virginia, is the contracting activity (HR0011-17-2-0042).
Source: Defense.gov, Darpa.mil