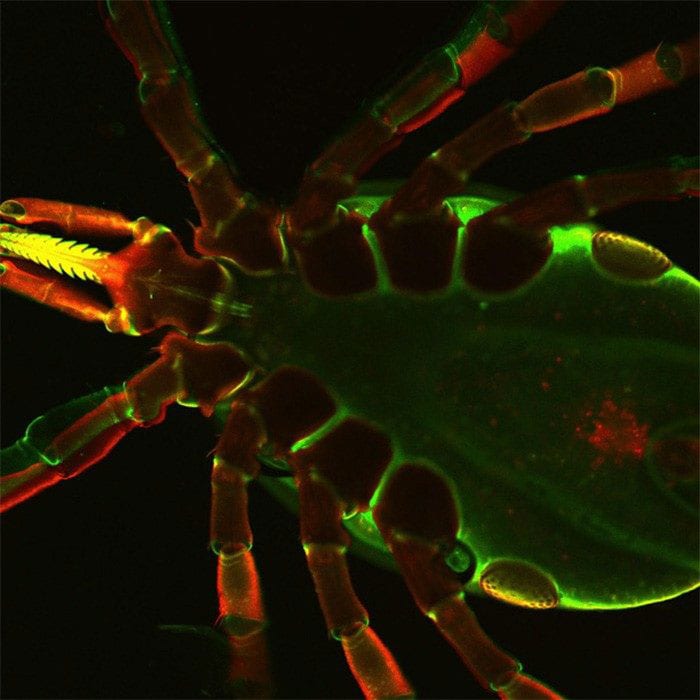
Dr. Utpal Pal wants to adapt the rabies vaccination platform to produce antibodies that can protect against Borrelia burgdorferi, the bacteria responsible for Lyme disease. The intention is to apply this work to other tick-borne diseases in the future.
“Borrelia infection is treatable with antibiotics, but it is often misdiagnosed,” explained Pal, professor in the Virginia-Maryland College of Veterinary Medicine’s Department of Veterinary Medicine at the University of Maryland College of Agriculture and Natural Resources. “A substantial fraction of antibiotic-treated Lyme patients develop a variety of neurological symptoms called post-treatment Lyme Disease Syndrome. There is no cure for that, no drug and no treatment. The only way to prevent this is to prevent Lyme, and a vaccine is a good answer for that.”
An expert in Borrelia and tick-borne disease, Pal is partnering with Matthias Schnell, director of Thomas Jefferson University’s Jefferson Vaccine Center, which is known for the study and application of the rabies virus as a platform for vaccination.
“We are using the rabies virus as a delivery platform to send in some vaccine candidates for Borrelia,” Pal said. “For rabies, we can produce an inactivated virus that helps the body produce the antibodies needed to fight it. Since we can produce the rabies vaccine and antibody proteins safely, why not have this virus produce other types of proteins that can do something else, like fight Borrelia?”
Although proven by Schnell and colleagues to be effective for other viral vaccinations, this approach has not yet been explored for Borrelia and tick-borne diseases.
Using proteins that Pal’s lab previously identified as vaccine candidates, Pal and his team hope to combine these proteins with the rabies virus to deliver long-lasting, safe, and effective immunity. Their work will include testing the four candidate proteins, along with three major types of rabies vaccine platforms that could be effective for Lyme disease.
Viral vaccines are produced in three different ways: live attenuated virus, which is rendered incapable of producing full-blown infection; inactivated virus, which is more popular because of its safety and efficacy and is the platform for the actual rabies vaccine; and virus-like particle, or VLP, which is created by extracting a protein from the virus and producing a shell with viral proteins only on the outside, tricking the body into developing defenses against the virus itself.
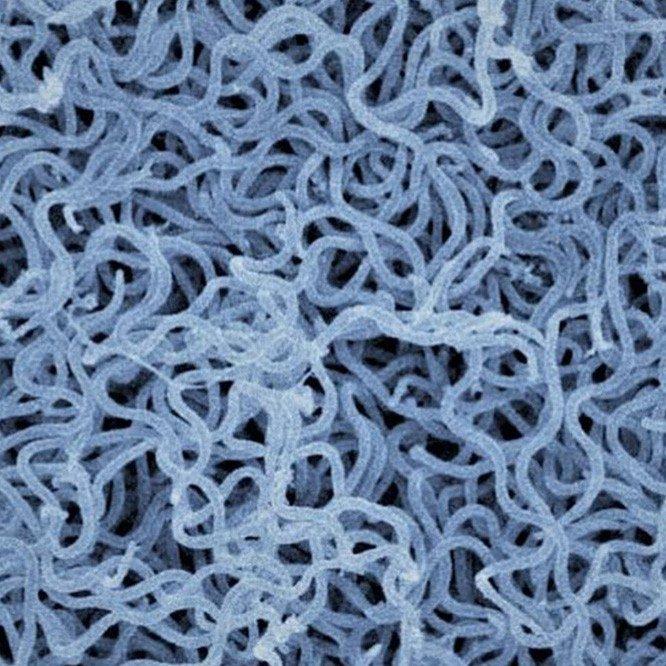
Pal and Schnell will examine all three of these possible platforms, using the rabies virus itself as the ultimate delivery method.
Because a target protein, or antigen, will simply disappear into the bloodstream if injected wholesale, a vaccine must include a delivery method. For that reason, vaccines use adjuvant chemicals, “a compound that makes the antigen stable so that the immune system can recognize it, see it, and mount an immune response,” Pal said.
The best adjuvants aren’t safe for humans, but the use of a virus platform eliminates the need for an adjuvant. “We are using the virus’ ability to produce a strong immune response to supply the Borrelia proteins and produce the appropriate antibodies,” explained Pal.
In addition to this unique approach, Pal is investigating the ability to stop Borrelia infection from two different angles, targeting both Borrelia proteins and the tick proteins that help keep the bacteria alive and ensure transmittal to humans.
“It is as if we are trying to dismantle the train [Borrelia] and the tracks [the delivery mechanism from ticks to humans] at the same time, which has never been done before,” Pal said. “Tick proteins are helping Borrelia, and with the vaccine, we would like to develop antibodies to try to prevent the infection so that when the tick starts to feed, those antibodies will bind to tick targets, as well as Borrelia.”
To accomplish this, Pal will target proteins previously identified by his lab, including two Borrelia proteins, BB0405 and BBI39 (The Journal of Infectious Diseases) and two parts of a tick protein (Journal of Biological Chemistry).
Once the best combination of vaccine platform and protein antigens has been identified, Pal and his team will test in mice to examine the vaccine’s underlying protection mechanisms and determine its overall effectiveness.
“This is an exciting project because we are able to repurpose the very effective rabies virus delivery platform to create something new,” Pal said. “Lyme disease is a very prevalent infection, but with a cure that doesn’t work for everyone. These chronic cases create long-term problems for diagnosis and treatment, and those patients are very difficult to manage. If we can produce something effective for Lyme disease, we can help with this problem and also understand how this solution can be applied to other tick-borne diseases.”