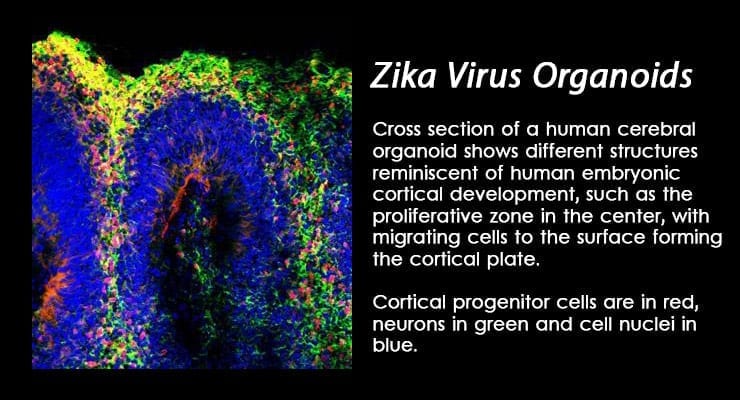
The Global Biodefense Zika Snapshot is a quick update on the latest news and research related to the Zika virus emergency.
World’s First Zika Virus Infectious cDNA Clone
A multidisciplinary team from The University of Texas Medical Branch at Galveston is the first in the world to genetically engineer a clone of the Zika virus strain, a development that could expedite many aspects of Zika research, including vaccine and therapeutics development.
Cloning the virus unlocks scientists’ ability to more quickly develop countermeasures and explore whether or how the Zika virus has evolved to spread more quickly and cause more severe diseases in people. This study is currently available in Cell Host and Microbe.
Furthermore, the team engineered a luciferase reporter Zika virus. Luciferase is the chemical in fireflies that gives them their signature glow. The “glowing” reporter virus could be used for antiviral drug screening. In addition, the reporter signal could be used to track Zika virus infection in mosquitoes and small animal models.
NSF Awards $1.7 Million in Rapid Response Grants to Study Zika Virus
The National Science Foundation (NSF) Division of Environmental Biology’s Ecology and Evolution of Infectious Diseases (EEID) Program has funded nine rapid response, or RAPID, grants totaling $1.7 million.
- David Abramson, New York University: The Risk Salience of Zika Virus Infection in an Environment of Shifting Scientific, Social, and Policy Uncertainty and Discourse
- Derek Cummings, University of Florida: Data driven mathematical modeling of the shared epidemiology of Zika and other arboviruses across the globe
- Nina Fefferman, University of Tennessee, Knoxville: Modeling Zika Control Effectiveness with Feedback in Risk Perception and Associated Demand across Scales of Intervention
- Courtney Murdock, University of Georgia: Environmental Drivers of Zika Transmission and Control
- Alex Perkins, University of Notre Dame: Overcoming Uncertainty to Enable Estimation and Forecasting of Zika Virus Transmission
- Michael Riehle, University of Arizona: Determining the Extrinsic Incubation Period (EIP) and Transovarial Transmission Potential of Zika Virus in Aedes aegypti mosquitoes
- Anna Stewart, SUNY, Upstate Medical University: In-situ Vector Dynamics in a High Burden Region in Ecuador
- Jefferson Vaughan, University of North Dakota Main Campus: Potential enhancement of Zika Virus Transmission by Microfilarial Nematodes
- Gonzalo Vazquez Prokopec, Emory University: Harnessing Spatial Heterogeneity to Contain Zika Virus Transmission
Experimental Drug Against Hepatitis C Slows Down Zika Virus Infection in Mice

Researchers from the KU Leuven Laboratory of Virology and Chemotherapy (Rega Institute) have published a study showing that an experimental antiviral drug against hepatitis C slows down the development of Zika in mice. The research team was led by Professor Johan Neyts.
“As the Zika virus is related to the hepatitis C virus, we examined whether some inhibitors of the hepatitis C virus also prevent the multiplication of the Zika virus in human cells. We have identified at least one experimental drug that is effective against the Zika virus.”
ZeptoMetrix Releases Additional Zika Virus Culture Fluids
ZeptoMetrix Corporation today announced the release of 3 additional Zika Virus Strains to its product inventory to aid in the rapid development of diagnostic tests and for the manufacture of quality control reagents to monitor their effectiveness.
“We are offering as much support as possible to assist during this global Zika virus emergency”, explains Dr. Gregory R. Chiklis, President and CEO of ZeptoMetrix. “Our focus, has always been and will continue to be to support the diagnostics industry with as many precise, dependable and non-infectious tools as we can.”
Study: Brazilian Zika Virus Strain Causes Birth Defects
Researchers at the UCSD School of Medicine, with colleagues in Brazil and Senegal, have published the first “direct experimental proof” that the Brazilian strain of Zika virus can actually cause severe birth defects.
“Rising infection rates of Zika virus in places like Brazil, with a corresponding increase in cases of microcephaly, have powerfully suggested an association, but until now hard evidence has been lacking,” said Alysson R. Muotri, PhD. “Our findings provide direct experimental proof that the Brazilian Zika virus strain causes severe birth defects – and that the full adverse effect upon health, even beyond microcephaly, is not yet fully understood.”
The data in mice also suggest that microcephaly is only the tip of the iceberg. The animals have extensive intra-uterine growth arrest, which essentially means poor fetal development in the womb. Media covering the Zika story have focused upon affected babies with small heads because such images are profoundly dramatic, but the true health impact is likely to be more widespread and devastating.
CDC Announces Funds for States and Territories to Prepare for Zika
The U.S. health department has made available more than $85 million to fight Zika infection in the country, the CDC said on Friday. More than $85 million in redirected funds identified by the Department of Health and Human Services is being made available to support efforts to protect Americans from Zika infection and associated adverse health outcomes, including the serious birth defect microcephaly.
“These funds will allow states and territories to continue implementation of their Zika preparedness plans, but are not enough to support a comprehensive Zika response and can only temporarily address what is needed,” said Stephen C. Redd, M.D. (RADM, USPHS), director of CDC’s Office of Public Health Preparedness and Response. “Without the full amount of requested emergency supplemental funding, many activities that need to start now are being delayed or may have to be stopped within months.”
Armed Forces Health Surveillance Branch Funds Studies On Zika Virus

The Armed Forces Health Surveillance Branch (AFHSB) has allocated $1.76 million in additional funding to 10 Department of Defense laboratories to enhance their surveillance efforts related to the Zika virus epidemic and assess the mosquito-borne disease’s current and potential impact on the health and readiness of U.S. military forces. Laboratories to receive funding include:
- The Naval Health Research Center, based in San Diego, Calif., will receive $204,600 to conduct surveillance in Department of Defense and civilian populations in Arizona, Texas and California.
- The Walter Reed Army Institute of Research based in Silver Spring, Md., will receive $186,000 to conduct clinic-based surveillance at the U.S. Army Garrison, Fort Buchanan, in Puerto Rico, where Zika is emerging, as well as to conduct a serosurvey for Zika among Department of Defense personnel in the Caribbean and other overseas locations.
- The Naval Infectious Diseases Diagnostic Laboratory at the Naval Medical Research Center in Silver Spring, Md., will receive $143,000 to support clinic-based surveillance in the southern Caribbean countries of Grenada, Trinidad and St. Lucia.
- The U.S. Army Medical Research Directorate-Kenya will receive $150,000 to conduct retrospective surveillance for Zika in humans and mosquitoes in Kenya.
- The Armed Forces Research Institute of Medical Sciences, based in Bangkok, Thailand, will receive $248,000 to conduct retrospective surveillance for Zika in humans and mosquitoes in Thailand.
- The U.S. Naval Medical Research Center- Asia, based in Singapore, will receive $230,340 to conduct retrospective surveillance for Zika in humans and mosquitoes in Bangladesh, Cambodia and Laos.
- The Naval Medical Research Unit No. 3, based in Cairo, Egypt, will receive $105,100 to expand current arthropod-borne virus surveillance efforts in Liberia.
- The Naval Medical Research Unit No. 6, based in Lima, Peru, will receive $250,00 to expand current clinic-based febrile illness surveillance activities in Central and South America.
- The Navy Environmental and Preventive Medicine Unit-2, based in Norfolk, Va., will receive $90,000 to expand entomological surveillance for Zika in Haiti.
- The Naval Medical Research Center in Silver Spring, Md., will receive 105,100 to conduct a serosurvey for Zika and other vector-borne pathogens among Department of Defense personnel deployed to West Africa.
The funds, provided by AFHSB’s Global Emerging Infections Surveillance and Response (GEIS) section, will examine the prevalence of Zika infections through clinic-based studies among military beneficiary and civilian populations who receive healthcare at facilities in the southeastern U.S., the Caribbean, Central and South America, West Africa and Southeast Asia. The laboratories will also conduct surveillance for Zika virus in mosquitoes collected through ongoing entomological surveillance activities.