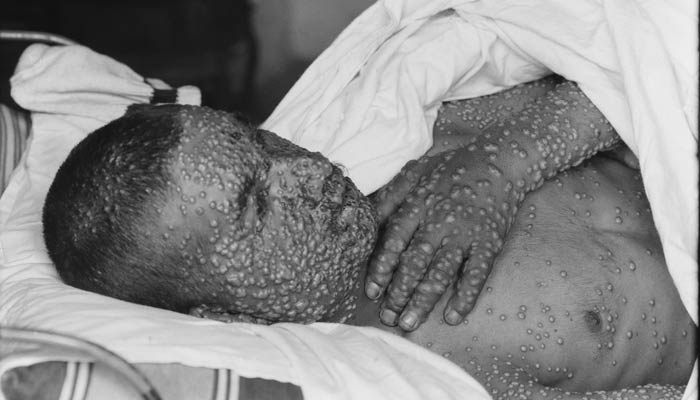
The FDA approved the Jynneos Smallpox and Monkeypox Vaccine last week. The live, non-replicating vaccine is made from a vaccinia virus, which is closely related to, but less harmful than, variola or monkeypox viruses and can protect against both of these diseases.
FDA Approves New Smallpox, Monkeypox Vaccine: What Is It For?
Smallpox used to be a huge problem around the world, killing an estimated 300 million people “in the 20th century alone.” About a third of those who got infected by the variola virus did not survive.
In fact, smallpox was so significant that it changed the course of history on many an occasion. For example, the virus may have helped the Spanish, British, and other European countries colonize the Americas. High school history books may not mention the fact that smallpox brought over by European settlers helped weaken and wipe out many of the Aztecs, Incas, and Native Americans who had been occupying the Americas and had never been previously exposed to the virus.
Indeed, not too long ago, smallpox was a major killer disease. That’s why the global eradication of smallpox that was declared complete in 1980 was such a big public health achievement. That’s also why any event that combines the words “accident” and “existing smallpox laboratory samples,” quickly results in worldwide news. This happened just last week when an explosion occurred in Vector, a Russian building that’s one of two facilities in the world housing the remaining samples of the smallpox virus. Fortunately, the explosion did not seem to affect the samples.
Read more by Bruce Y. Yee at Forbes
RELATED READING
Smallpox Is A Dormant Volcano
Vopr Virusol
The presence of rodent-associated natural foci containing at least 6 of the known 11 viruses belonging to the genus Orthopoxvirus (Poxviridae, Chordopoxvirinae) within the equatorial, tropical, subtropical, temperate, and subarctic climatic zones; the increasing aggravation of the monkey pox epidemic situation in equatorial Africa with an increase in human mortality by an average of 9.8% with a possibility of 2 to 8 passages in 30-70% of patients; the possible persistence of a virus in the human cadavers buried in the permafrost of Eurasia and America; bioterrorism threat due to the unaccounted viral reserves persisting somewhere or somebody; no postvaccinal human immunity since vaccination and vaccine manufacture stopped 30 years ago as recommended by the WHO, make the risk of the deteriorating epidemic situation with disastrous effects greater now and in the foreseeable future than it was 20-30 years ago.
Is Smallpox a Hazard in Church Crypts?
British Journal of Industrial Medicine
Although the risk is remote, the WHO and the UK Advisory Committee on Dangerous Pathogens (ACDP) informed HSE that, although the risk was remote, survival of virus on the preserved skin of a smallpox victim interred in a crypt over 100 years ago could not be excluded. The survival time of smallpox virus in preserved infected scabs has not been adequately documented, but viable virus was isolated from scabs kept in envelopes within a laboratory cupboard for up to 13 years when testing was discontinued.
Most authorities believe that in the presence of moisture – for example, in earth burials – smallpox virus is unlikely to remain viable for long, and no more than one to two years even in exceptional conditions. Survival of the virus within scabs on the preserved skin of smallpox victims buried in permafrost or in dry cool crypts, however, could be much longer.
Six Vials of Smallpox Discovered in U.S. Lab (2014)
Science
Federal scientists last week discovered a half-dozen forgotten vials of smallpox virus while cleaning out a storage area on the campus of the National Institutes of Health (NIH) in Bethesda, Maryland. The six vials of freeze-dried virus, apparently dating from the 1950s, were found by a scientist from the Food and Drug Administration (FDA) on 1 July in a cold storage room that was originally part of an NIH laboratory, but was transferred to FDA in the early 1970s.