Pfenex Inc. this week announced positive immunogenicity and safety data from the Day 70 analysis of the Px563L anthrax vaccine study. The results demonstrate tolerable safety and significant immunogenicity after only 2 doses of Px563L.
The randomized, double-blind, placebo-controlled phase 1a study enrolled 3 cohorts (10 mcg, 50 mcg and 80 mcg) in a dose-escalating manner. Within each cohort, subjects received Px563L, RPA563 or placebo in an 8:8:2 ratio.
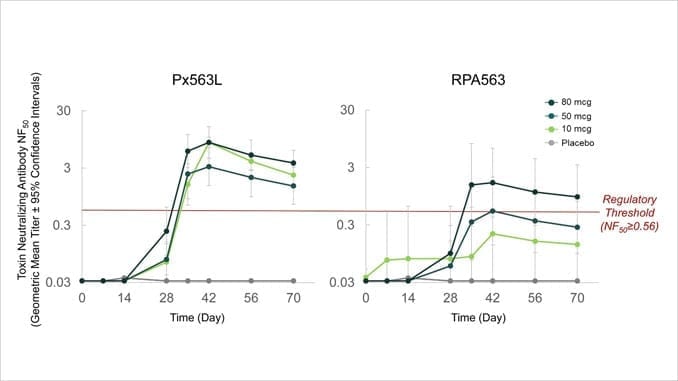
Subjects were administered 2 doses of vaccine or placebo 28 days apart. The current analysis covers safety and immunogenicity assessment, including toxin-neutralizing antibody (TNA) expressed as 50% neutralizing factor (NF50), through Day 70. TNA NF50 threshold value > 0.56 has been correlated with significant survival after anthrax exposure in animal models.
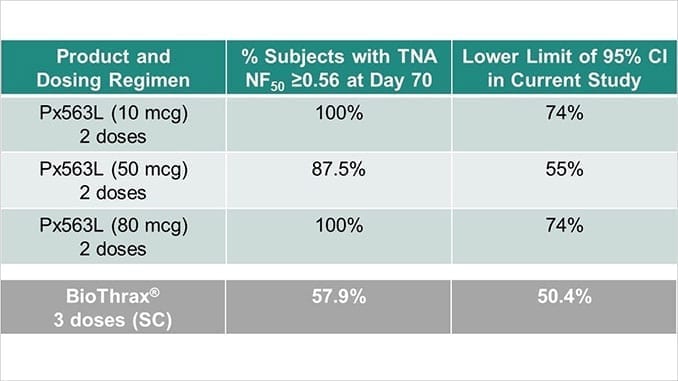
After administration of 2 doses, 100% of Px563L subjects achieved a TNA NF50 > 0.56 at Day 70 for both the 10 mcg and 80 mcg dose cohorts, and 87.5% achieved the target threshold in the 50 mcg dose cohort. For comparison, after subcutaneous administration of 3 Biothrax® doses (currently approved anthrax vaccine), 57.9% of subjects achieved a TNA NF50 >0.56 at Day 70 (Vaccine 2014; 32:2217-2224).
“The Px563L data from the Day 70 analysis of the phase 1 study are very encouraging, with the vaccine being well-tolerated and affording potentially superior protection after only 2 doses,” noted Dr. Hubert Chen, chief medical officer of Pfenex. Bertrand C. Liang, chief executive officer added, “We look forward to continuing the collaborative development of Px563L with BARDA and progressing towards a potential procurement of our next-generation anthrax vaccine to satisfy Strategic National Stockpile needs.
An additional criterion for assessing anthrax vaccine immunogenicity success is for the lower confidence limit (LCL), or the lower bound of the two-sided 95% confidence interval for the percentage of subjects who met or exceeded the TNA threshold (TNA NF50 > 0.56), to be greater than or equal to 40%.
 All doses of Px563L were above the LCL at Day 70, having demonstrated this activity after only 2 doses (10 mcg: 74%, 50 mcg: 55%, 80 mcg 74%). For comparison, Biothrax reported a LCL of 50.4% at Day 70 (Vaccine 2014; 32:2217-2224) following a three-dose regimen.
All doses of Px563L were above the LCL at Day 70, having demonstrated this activity after only 2 doses (10 mcg: 74%, 50 mcg: 55%, 80 mcg 74%). For comparison, Biothrax reported a LCL of 50.4% at Day 70 (Vaccine 2014; 32:2217-2224) following a three-dose regimen.
 Only mild Grade 1 injection site reactions were observed at the 10 mcg and 50 mcg Px563L doses as well as for all RPA563 doses. At the 80 mcg Px563L dose, in addition to Grade 1 findings, there were two Grade 2 reports of erythema (redness). There were no other Grade 2 or higher injection site reactions, specifically injection site pain, arm motion limitation, tenderness or swelling.
Only mild Grade 1 injection site reactions were observed at the 10 mcg and 50 mcg Px563L doses as well as for all RPA563 doses. At the 80 mcg Px563L dose, in addition to Grade 1 findings, there were two Grade 2 reports of erythema (redness). There were no other Grade 2 or higher injection site reactions, specifically injection site pain, arm motion limitation, tenderness or swelling.
Pfenex has leveraged its Pfēnex Expression Technology® platform to build a pipeline of product candidates and preclinical products under development including other biosimilars, as well as vaccines, therapeutic equivalents to reference listed drug products, and next generation biologics.