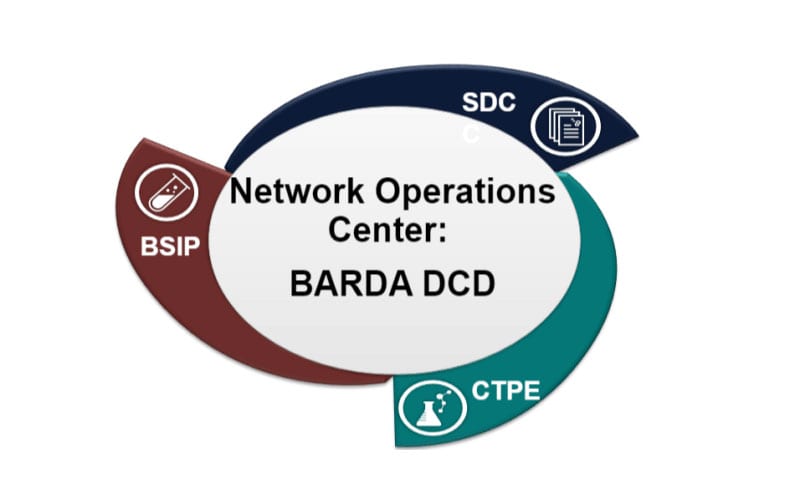
New contract awards by the Biomedical Advanced Research and Development Authority (BARDA) will support three integrated components of the Medical Countermeasures Clinical Studies Network (CSN) to provide federal partners and medical countermeasure developers with technical and regulatory assistance in the development of vaccines, therapeutics, diagnostics, and medical devices to prevent or treat the medical consequences of chemical, biological, radiological, and nuclear (CBRN) threats, pandemic influenza, and emerging infectious diseases.
BARDA established the CSN as a core service in 2012 and awarded IDIQ contracts to five full service clinical Contract Research Organizations in 2014 to form the CSN infrastructure. During the network’s five years, several important gaps in capabilities were identified. Three separate and distinct solicitations have been awarded to address the following gaps: Clinical Trial Planning & Execution services on a routine basis to address the requirements under regular non-emergency timelines, as well as services during emergencies when rapid timelines are crucial; a Biological Specimen and Investigational Product storage facility; and a Statistical and Data Coordinating Center.
The Clinical Trials Planning and Execution (CTPE) component will be responsible for planning and execution of clinical studies to support medical countermeasure development and was awarded to four recipients:
- ICON Government & Public Health Solutions
- Pharm-Olam LLC
- PRA Health Sciences, Inc.
- Technical Resources International, Inc.
The Statistical Data Coordinating Center (SDC) will serve to standardize data collection, management, and analysis across studies that use this BARDA-supported network and was awarded to:
The Biological Specimen and Investigational Product Storage Facility (BSIP) will provide a single facility for long-term storage of clinical study specimens and investigational products across BARDA-supported studies was awarded to: